- feeds
- popular
- recent
- reader
- about
- story
- technologies
- what is rss
- connect
- dmca
- contact
- Feed Preview mddi
Boston Scientific's Farapulse Is a Showstopper
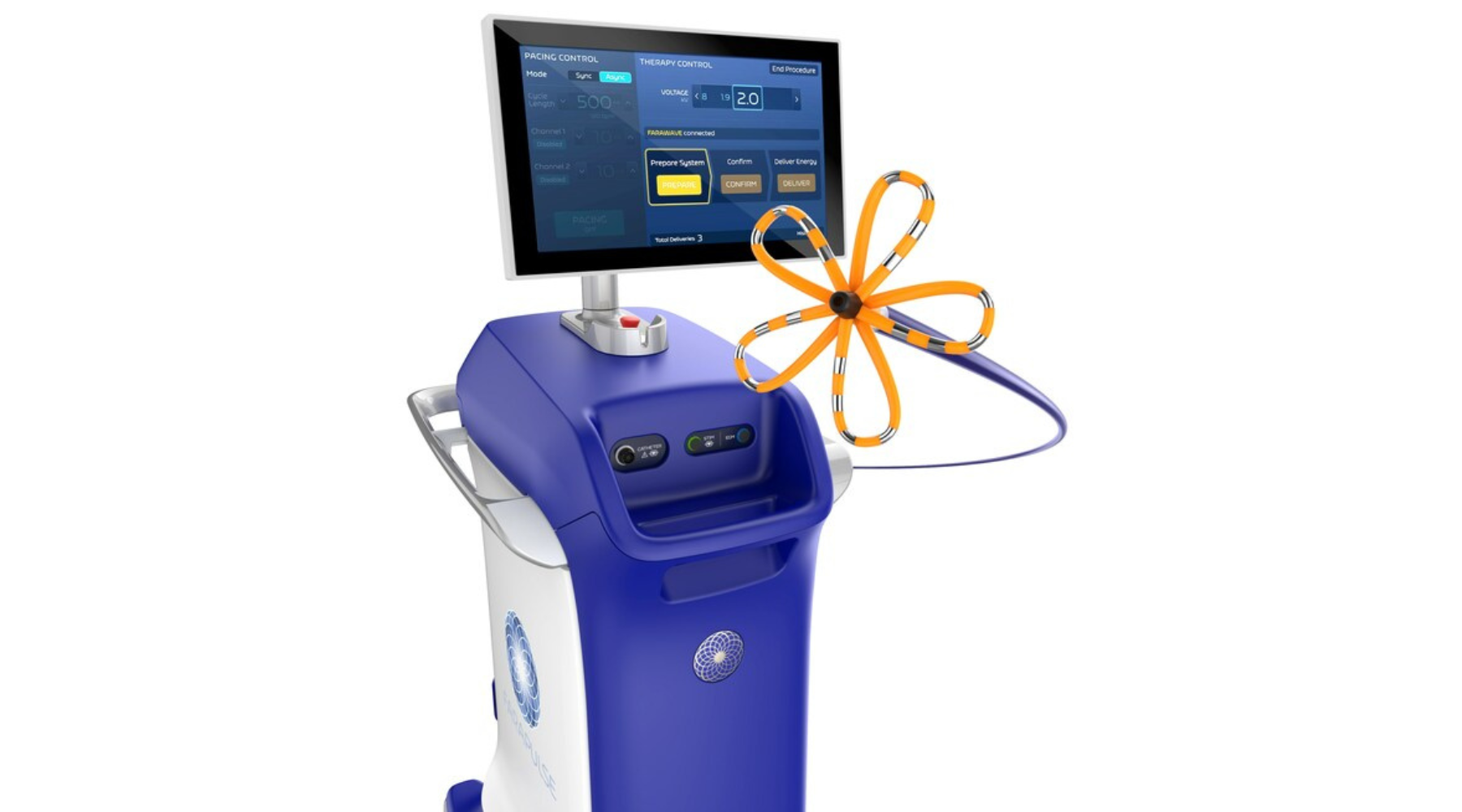
Electrophysiologists are giving the new pulsed field ablation system a warm welcome.
https://www.mddionline.com/cardiovascular/boston-scientific-s-farapulse-is-a-showstopper
How to Optimize Your Medical Injection Molding Process

To consistently mold components that meet quality control specs and reduce development hours for customers, follow these steps.
https://www.mddionline.com/manufacturing/how-to-optimize-your-medical-injection-molding-process
These Antibacterial Bioplastics Will Make You Say, ‘What the Cluck?’

Trivia Tuesday: Why did researchers take an interest in chicken eggs?
Exo’s FDA Cleared Cardiac & Lung AI Now on Iris Handheld Ultrasound
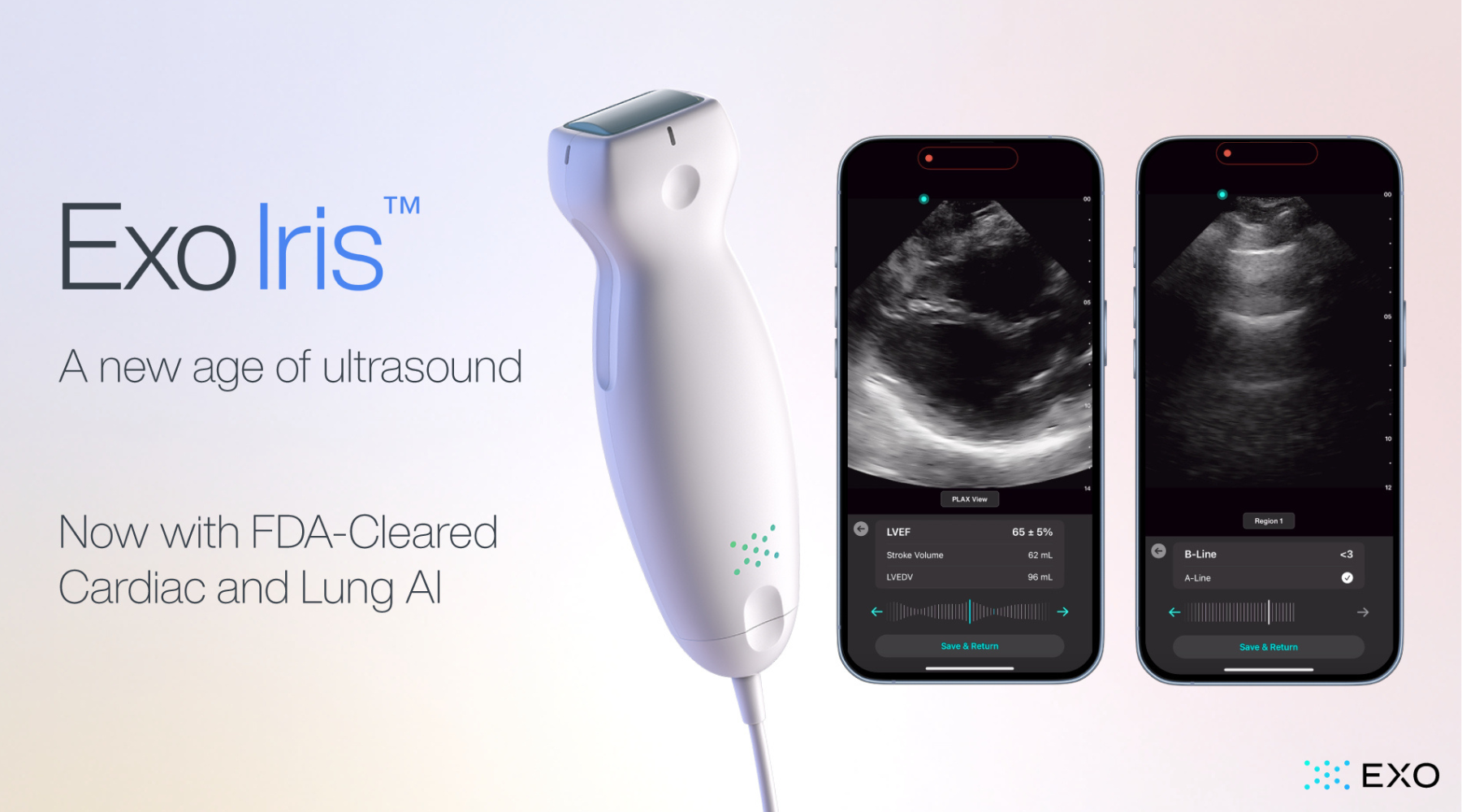
With the newly approved applications, Exo is now cleared for cardiac, lung, bladder, hip, and thyroid.
The New Frontier of Early-Stage Medical Device Funding

The rise of family offices and incubators is transforming the funding landscape for early-stage medtech companies, providing new opportunities and insights for success.
https://www.mddionline.com/startups/the-new-frontier-of-early-stage-medical-device-funding
NanoString's Assets to be Acquired by Bruker

In a year that’s already filled with M&A, Bruker said it will acquire NanoString for $392 million.
https://www.mddionline.com/ma/nanostring-goes-from-filing-bankruptcy-to-being-acquired
Breast Cancer Detection Drug-Device System Nabs FDA Approval
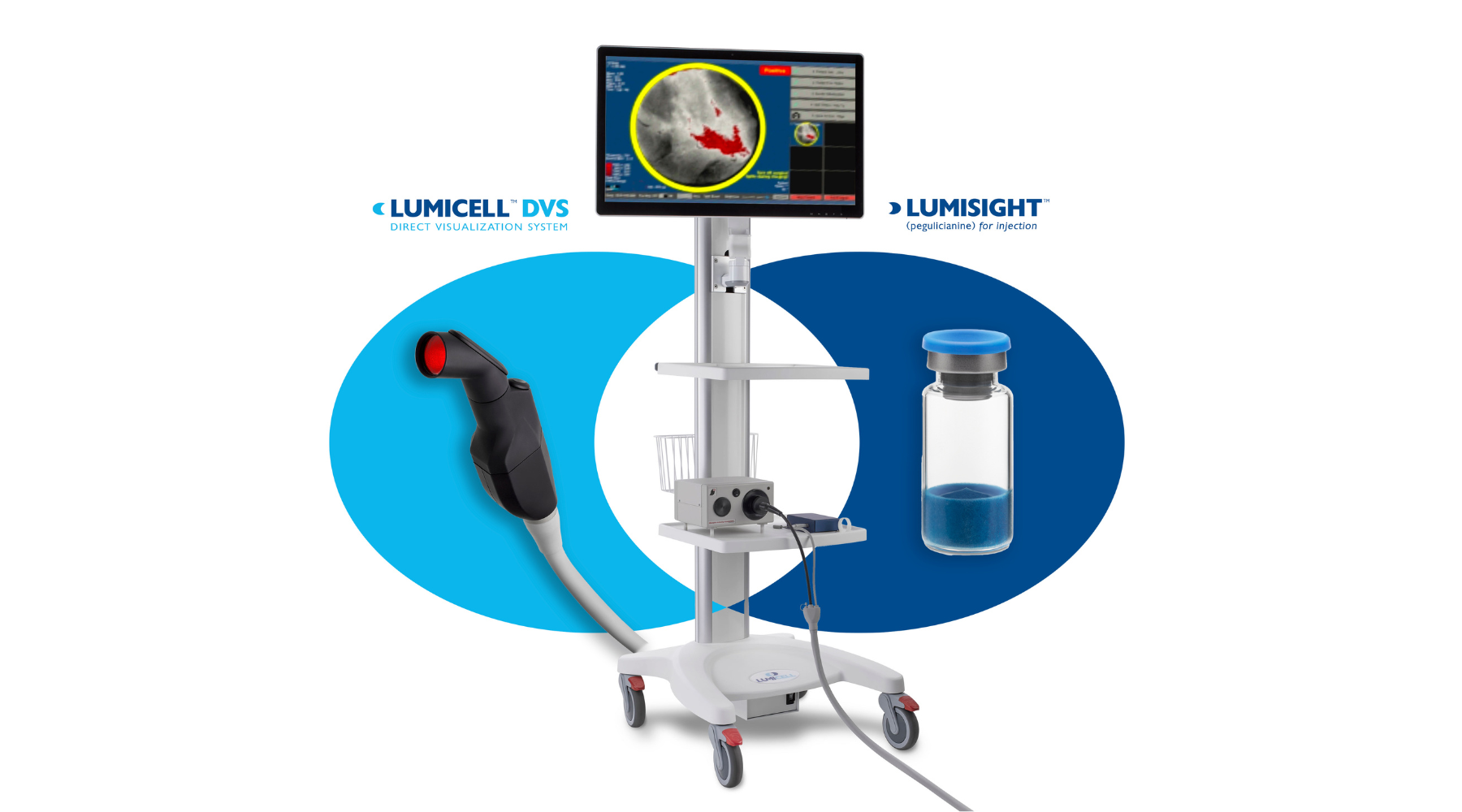
The LumiSystem enables surgeons to scan the breast cavity post-lumpectomy in real time, detect, and resect residual cancer that otherwise could have been missed.
DermTech Cuts 100, Seeks Strategic Alternatives

Over the past year, the company has been trying to stabilize its financial situation, including previous restructuring plans.
https://www.mddionline.com/business/dermtech-cuts-100-seeks-strategic-alternatives
Cool Device, Bruh, But Does It Work?

This week in Pedersen's POV, our senior editor weighs in on an evolving debate in medical device design.
https://www.mddionline.com/design-engineering/cool-device-bruh-but-does-it-work-
How Will Stryker Enter the Surgical Robotics Fray?

A medtech analyst looks at how the Kalamazoo, MI-based company could potentially enter the surgical robotics market.
https://www.mddionline.com/robotics/how-will-stryker-enter-the-surgical-robotics-fray-
Intuitive’s Throne Is Secure with New da Vinci 5

The new robot launch will make it harder for competitors to dethrone the robotics king.
https://www.mddionline.com/robotics/new-da-vinci-launch-could-catalyze-robotic-surgery-adoption
RSNA & GE HealthCare Collab to Bring Advanced Mammography Technology to Tanzania

The largest public hospital in the country will receive the latest mammography tech and hands-on training to improve breast cancer detection, diagnosis, and treatment.
Medtech in a Minute: US Authorities Throw the Book at Philips, and More

The medtech news you need. In one minute or less.
Formlabs Releases Fastest 3D Printer Yet

The Form 4 can achieve vertical print speeds of 100 mm per hour and outpace injection molding, according to the company.
https://www.mddionline.com/3d-printing/formlabs-releases-fastest-3d-printer-yet
Porex Partners With UK-based Diagnostics Startup

The partnership will scale up production of ReadyGo Diagnostics’ signature device and give it global reach.
https://www.mddionline.com/ivd/porex-partners-with-uk-based-diagnostics-startup
Fresenius Kabi USA Infusion Pump Software Recall Declared Class I
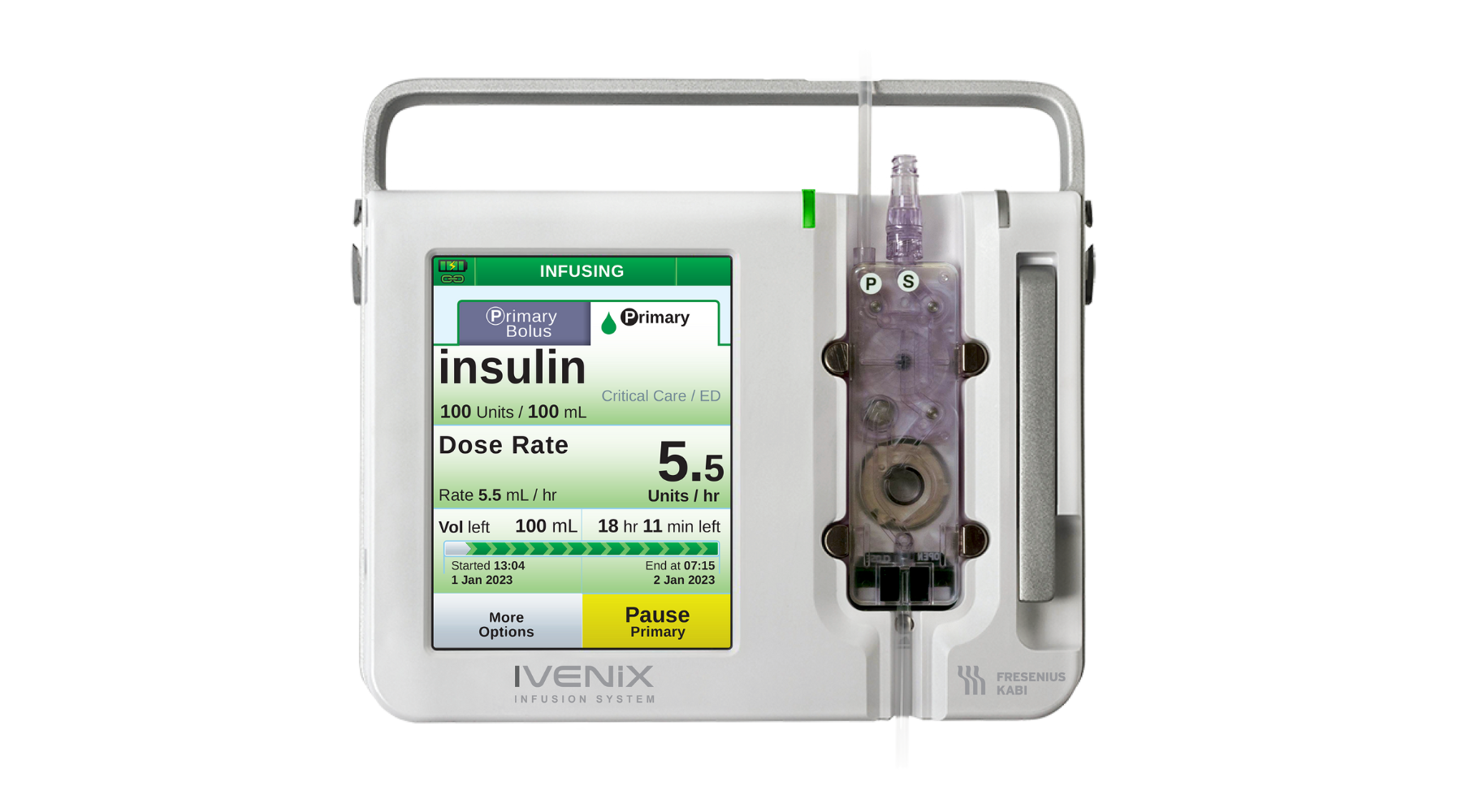
The company is urging customers to update the device software in order to mitigate noted software anomalies.
Abbott Has a Powerful Product Lineup in the Chamber
The company is on track to be the fastest-growing company in medtech again this year.
https://www.mddionline.com/business/abbott-has-a-powerful-product-lineup-in-the-chamber
Ensuring Today’s Innovations Do Not Become Tomorrow’s Problems

Consider these tips for engineering sustainability into products and processes without compromising performance and safety.
Vivasure Opens up about Securing a Top Spot in Vessel Closure Market
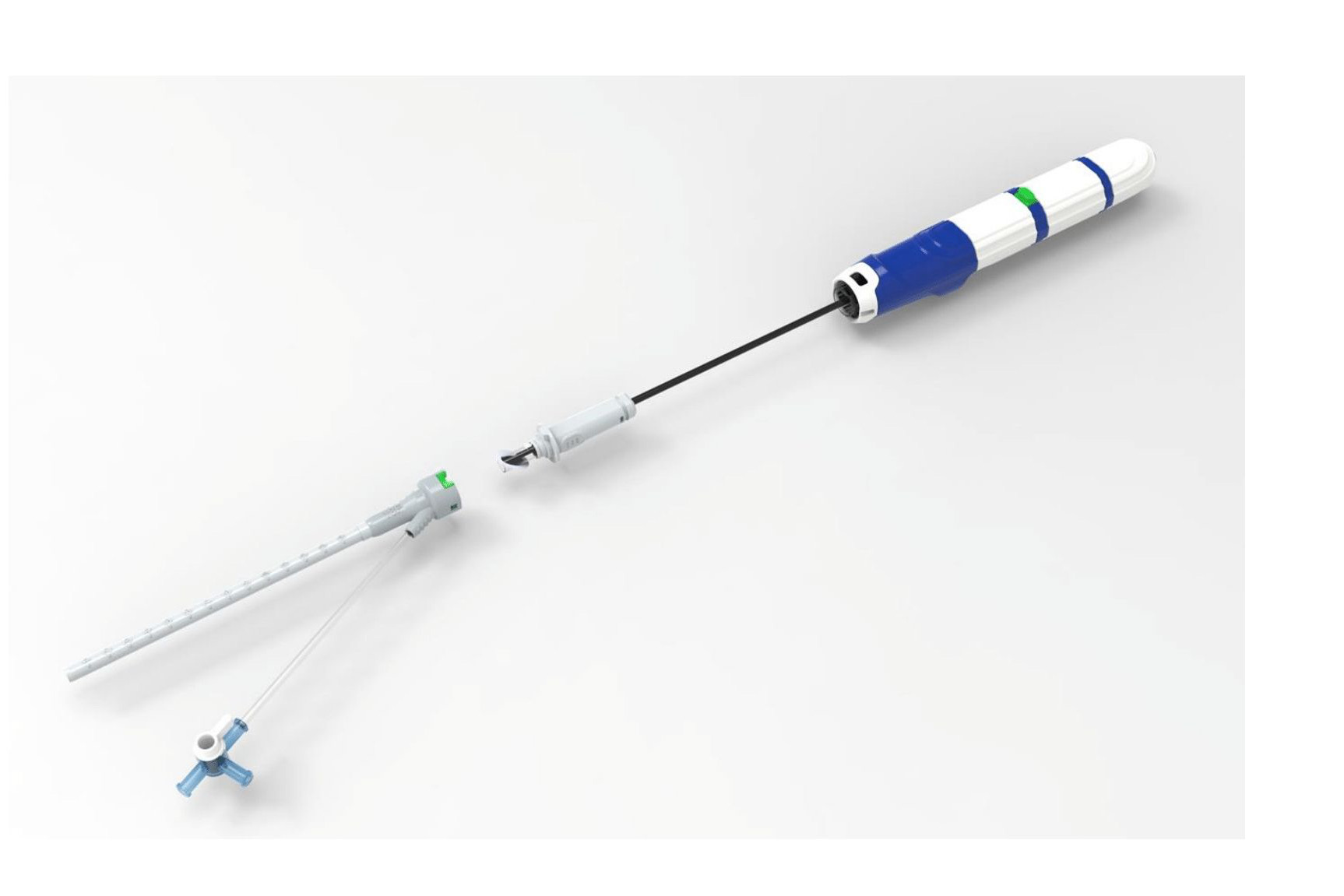
Recent approvals in the TAVR and TMVR markets are providing significant opportunities for companies like Vivasure Medical. The Galway, Ireland-based company recently said the first large-bore ven...
Boston Scientific Class I Recall Linked to Two Deaths

Categorized as a correction, not product removal, the company is recommending physicians not use the aliquot technique to deliver the Obsidio Conformable Embolic due to increased patient risk.
https://www.mddionline.com/regulatory-quality/boston-scientific-class-i-recall-linked-to-two-deaths
Hyperfine Enrolls First Patients in Swoop CARE PMR Study

The study will evaluate whether portable MR brain imaging can reliably identify brain swelling and bleeding associated with Alzheimer’s amyloid-targeting therapy at the same time as infusions t...
https://www.mddionline.com/neurological/hyperfine-enrolls-first-patients-in-swoop-care-pmr-study
Are More Deals in J&J's Future?

The company's chairman and CEO hinted at the possibility of more M&A during a recent earnings call.
A Better AI Tool for Diagnosing & Assessing Autism

EarliTec's CTO shares how the company’s autism diagnostic and assessment technology makes diagnosing autism easier for clinicians and families while avoiding common AI pitfalls.
https://www.mddionline.com/artificial-intelligence/a-better-ai-tool-for-diagnosing-assessing-autism
What Digital Health Startup’s Need to Know About the EU AI Act

The act lays down the law for artificial intelligence development and usage, dishing out hefty fines and a laundry list of must-dos for outfits dabbling in AI.
3D-printed PEEK-based Cranial Implants Cleared by FDA

The VSP PEEK Cranial Implant system includes a complete FDA-cleared workflow comprising segmentation and 3D-modeling software, a printer developed by 3D Systems, and PEEK resin from Evonik.
https://www.mddionline.com/implants/3d-printed-peek-cranial-implants-cleared-by-fda
14 Deaths Connected to Latest Abbott HeartMate Recall

Long-term buildup of biomaterials can obstruct the device, making it less effective at helping the heart pump blood.
https://www.mddionline.com/qa-qc/14-deaths-connected-to-latest-abbott-heartmate-recall
Roche Lands Breakthrough Designation for Alzheimer’s Test
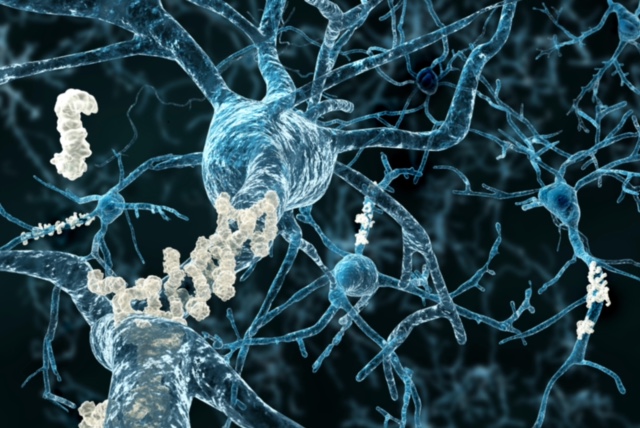
The Basel, Switzerland-based company’s test looks for the presence of amyloid pathology in patients suspected of having Alzheimer’s.
https://www.mddionline.com/ivd/roche-lands-breakthrough-designation-for-alzheimer-s-test
Fueling the Fire of Access to Emerging Medical Devices

This week in Pedersen's POV, our senior editor pushes for patient access to breakthrough medical devices.
How to be a Packaging Visionary in Pharma and Medtech

Here’s how you can be the leader you’ve always wanted to be, even within the prescribed limits of industry regulations.
https://www.mddionline.com/packaging/how-to-be-a-packaging-visionary-in-pharma-and-medtech
Bridging the Gap Between Research & Medical Device Development

In this Q&A, MD+DI investigates the connection between how better collaboration among academic institutions and industry could improve the production of medical devices.
https://www.mddionline.com/rd/bridging-the-gap-between-research-medical-device-development
FemInnovation CEO to Keynote IME South

Bethany Corbin, a recognized thought leader in women's health, is set to keynote IME South. The six-in-one advanced design and manufacturing expo returns June 4-6 to the Charlotte Convention Cent...
EU Regulators Approve Illumina’s Plan to Divest Grail

Illumina hopes to finalize the plan with EU regulators sometime in 2Q24.
https://www.mddionline.com/ivd/eu-regulators-approve-illumina-s-plan-to-divest-grail
Behind the Design: Form Follows Function in New L-Shaped Widex Hearing Aid

A Widex product designer shares insights behind the design of the company's newest hearing aid.
Philips Faces Consent Decree that Restricts Sales of New Sleep Apnea Devices

The decree restricts manufacturing and distribution of devices at or from Philips Respironics facilities in PA and CA until it has completed repair, rework, replacement, and refund activities.
Is the SMART Trial a Game Changer for TAVR?

Analysts discuss the potential impact of a clinical trial that had Medtronic’s Evolut technology go head-to-head with Edward’s Sapien platform.
https://www.mddionline.com/cardiovascular/is-the-smart-trial-a-game-changer-for-tavr
There’s A New Surgical Robotics Solution in Town

Medical Microinstruments reported it has received a De Novo Classification for its Symani Surgical System.
https://www.mddionline.com/robotics/there-s-a-new-surgical-robotics-solution-in-town
New Cardiovascular Company Emerges from Stealth Mode

Fresh off a $42 million series A financing, Reprieve Medical is developing an intelligent automated fluid management system for heart failure patients.
https://www.mddionline.com/startups/new-cardiovascular-company-emerges-from-stealth-mode
Medical Injection Molder Schnipke Acquired by Atalys

The acquisition will enhance its manufacturing capabilities, technology, and geographical footprint, said Atalys.
https://www.mddionline.com/business/medical-injection-molder-schnipke-acquired-by-atalys
6 Trendsetting Companies in the Cardiovascular Market

The cardiovascular market is one of the hottest segments in the medical device industry. MD+DI looks at the companies making the most noise in cardiology.
https://www.mddionline.com/cardiovascular/6-most-influential-cardiovascular-companies
Apple Asks Appeals Court to Reverse Smartwatch Import Ban as Masimo Patent Battle Continues

The tech giant said the decision to ban the watches were made relying on Masimo patents that were “purely hypothetical” when filing the complaint in 2021.
A Drowning Medtech Company Clings to a Life Preserver

Asensus Surgical was once poised to take on Intuitive Surgical. Now the company clings to a life preserver from a prospective buyer.
https://www.mddionline.com/ma/a-drowning-medtech-company-clings-to-a-life-preserver
New Approach, Automated Lab Streamlines Battery Chemistry Testing

University of Michigan research showcases how automated laboratories expedite chemical testing, enabling transformative battery advancements.
Medos’ Cerenovus Cerebase DA Guide Sheath Recall Categorized as Class I

The recall comes after reports of cracking at the distal catheter shaft, possibly resulting in patient injury.
Medtech Mega Mergers: Cautionary Tale or Compelling Growth Strategy?

This week in Pedersen’s POV, our senior editor revisits the trend of “biggering” in the medical device industry through a new lens.
https://www.mddionline.com/ma/medtech-mega-mergers-cautionary-tale-or-compelling-growth-strategy-
Medtech in a Minute: Abbott's Big FDA Approval, and More

The news you need. In one minute or less.
https://www.mddionline.com/cardiovascular/medtech-in-a-minute-abbott-s-big-fda-approval-and-more
High-Profile Merger Hits a Regulatory Roadblock

An FTC request stalls Boston Scientific's acquisition of Axonics.
https://www.mddionline.com/ma/high-profile-merger-hits-a-regulatory-roadblock
J&J Puts the Rumors to Bed & Moves to Acquire Shockwave

The New Brunswick, NJ-based company could strengthen its position in the cardiovascular intervention market with this $13.1 billion acquisition.
https://www.mddionline.com/ma/j-j-puts-the-rumors-to-bed-moves-to-acquire-shockwave
M&A & New Hires Dominate Medical Device Supplier News

Keeping you up to date on all the medical device supplier news.
https://www.mddionline.com/ma/new-hires-m-a-dominate-medical-device-supplier-news
Smith Medical Recalls PneuPac paraPAC Plus 300 and 310 Ventilator Kits

The recall reported the possibility of the devices producing continuous positive gas flow instead of the intended cycling when switched to the ventilate operating mode.
Teleflex, Arrow International Recall Arrow QuickFlash Radial Artery and Radial Artery/Arterial Line Catheterization Kits

The kits are being recalled after reports of increased resistance in the guidewire handle and chamber during use.
